

28 Aug
Why Are Methanol and Acetonitrile the Most Widely Used Solvents in RP-HPLC?
In RP-HPLC, which commonly employs C18, C8, C4, and phenyl columns, the choice of mobile phase solvents is a critical factor in achieving accurate, reproducible, and reliable analytical results. In the fields of food, pharmaceuticals, and environmental analysis, ensuring precision and consistency is part of our daily mission. Among the solvents available, methanol (MeOH) and acetonitrile (ACN) are predominantly used.

☞ Why these two?
-
Polarity and compatibility:
Both MeOH and ACN are polar organic solvents, fully miscible with water, making them versatile for RP-HPLC. This compatibility allows their use in both gradient and isocratic elution modes, depending on the polarity of the analyte. -
Elution strength:
ACN provides higher elution strength, enabling faster separations with sharper peaks, which enhances overall analysis quality. Methanol, although weaker in elution power, offers better selectivity for certain analytes and is more cost-effective for routine analysis. -
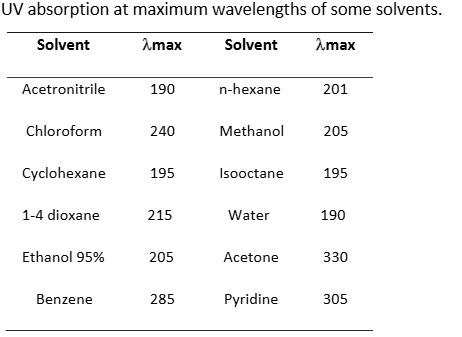
Low UV absorbance:
-
ACN: ~190 nm
-
MeOH: ~205 nm
This makes them highly suitable for UV-based detections. For example, compounds like aldehydes, ketones, citric acid, and vitamin B7 absorb in the 205–215 nm UV range, where ACN is more effective than MeOH. Conversely, methanol can be advantageous for chlorinated organics, pigments, fat-soluble vitamins, and polyaromatic compounds.
-
-
Viscosity:
Lower viscosity reduces backpressure in the HPLC column, ensuring stable flow rates. ACN has lower viscosity than MeOH, making it more suitable for long runs or when working with delicate columns. -
High purity:
Both solvents are available in HPLC-grade quality, minimizing baseline noise and making them ideal for both qualitative and quantitative analyses.



